CARTOX
Published in academic literature
App Summary
App Screenshots





Detailed Description
Functionality & Mechanism
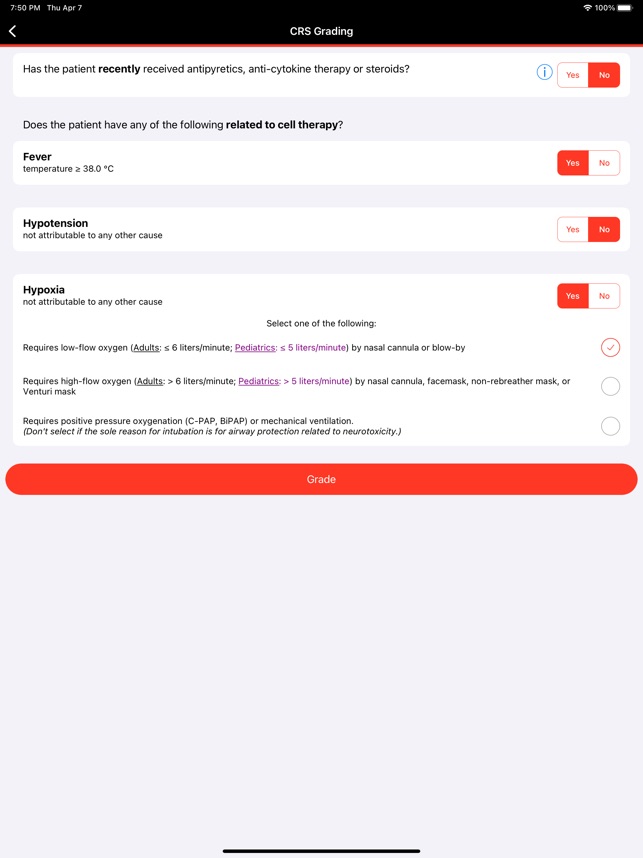
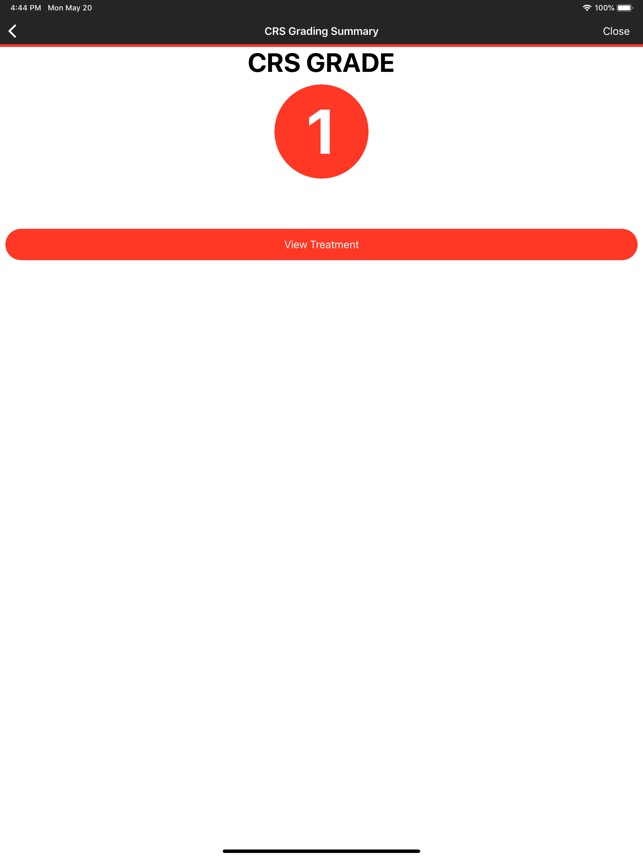
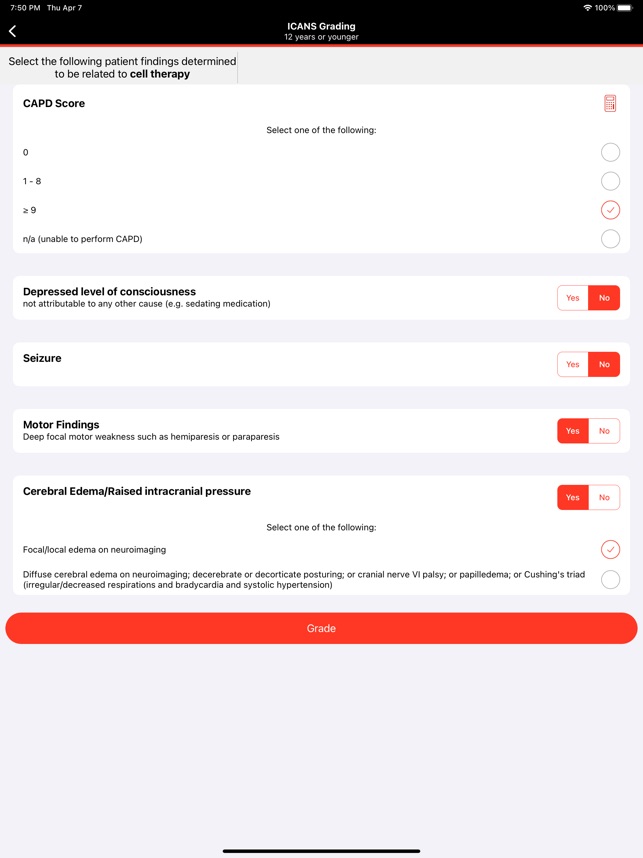
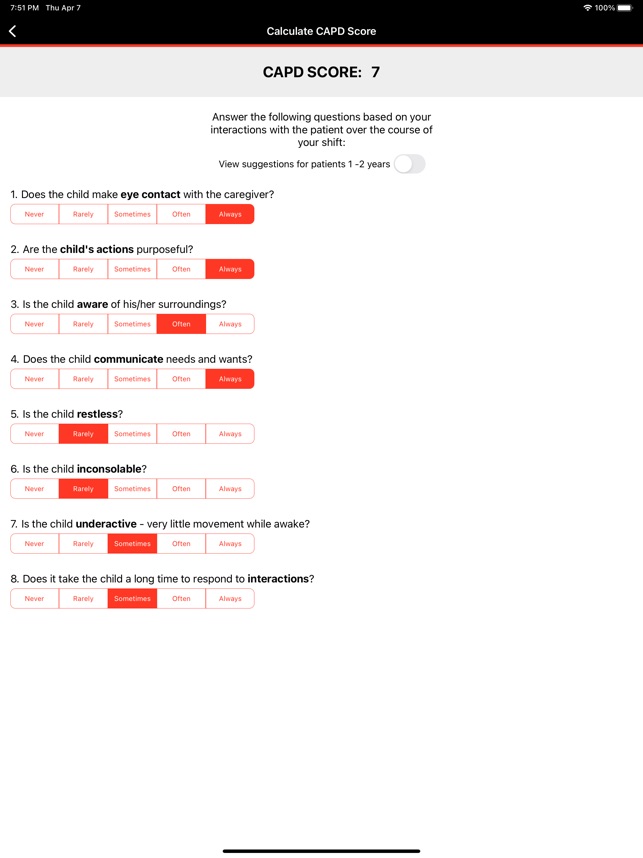
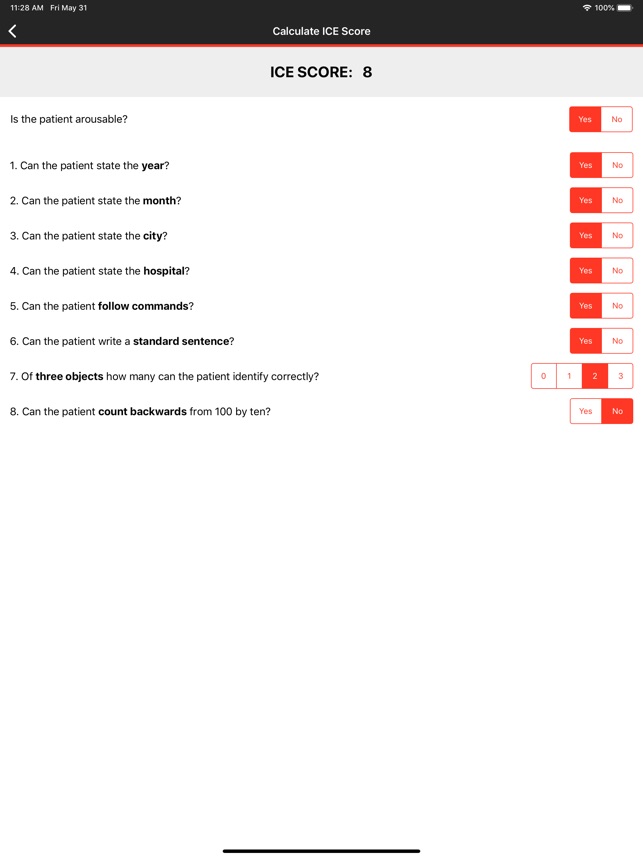
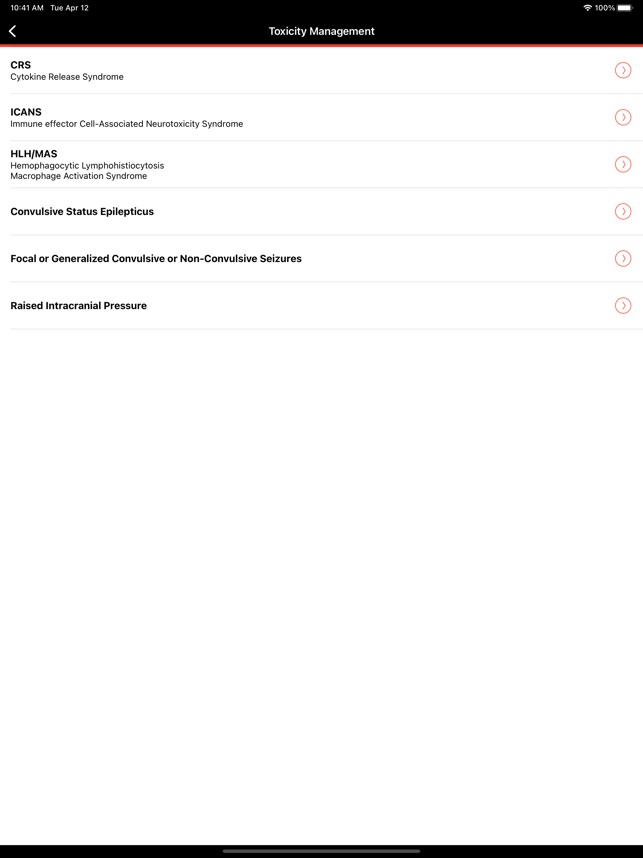
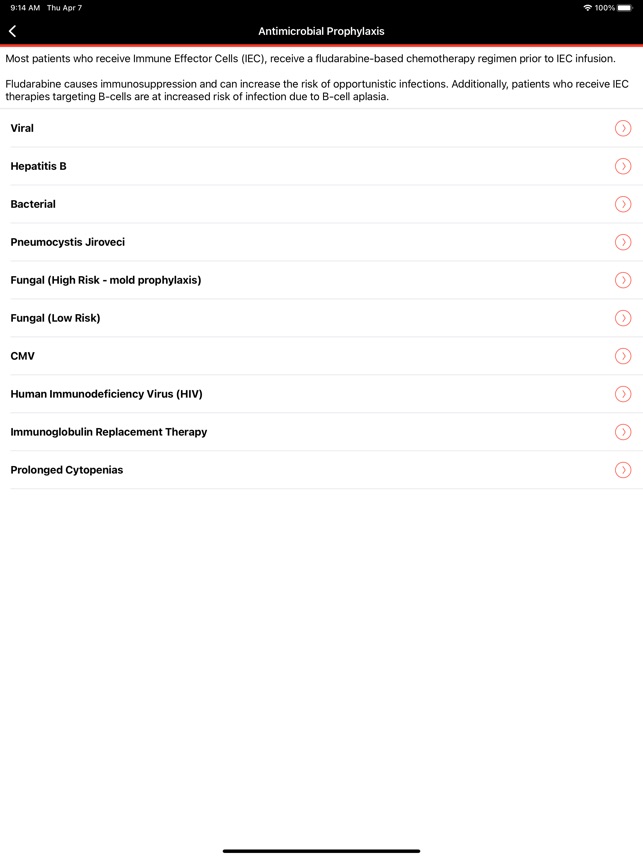
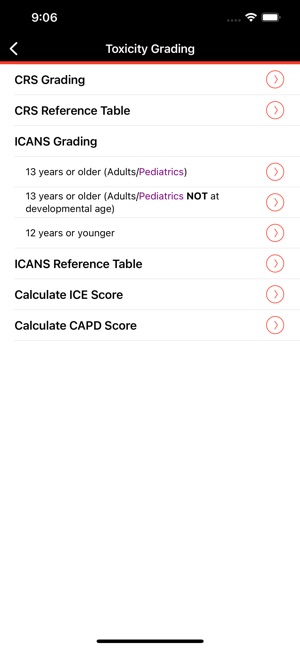
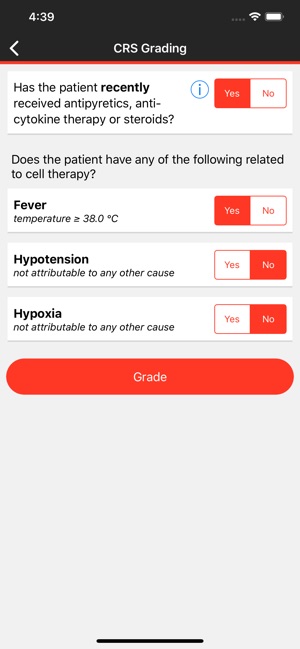
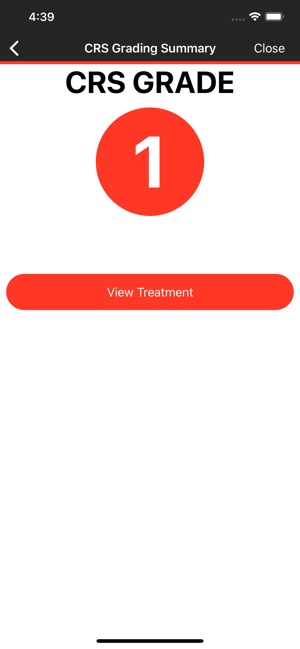
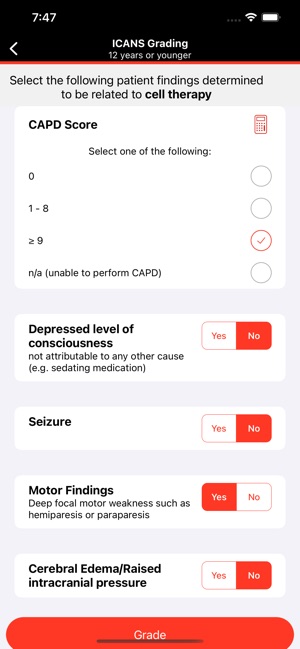
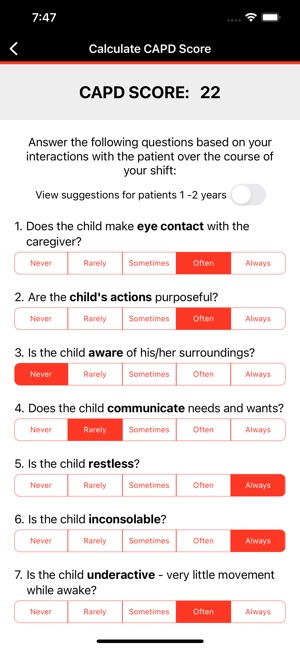
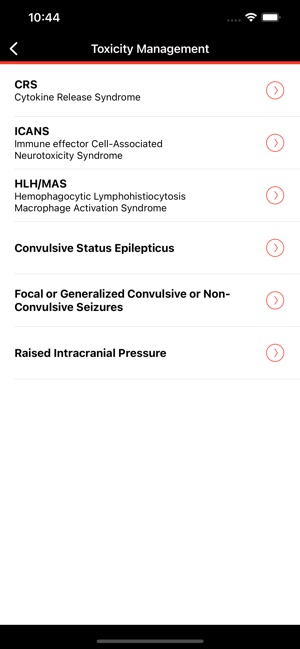
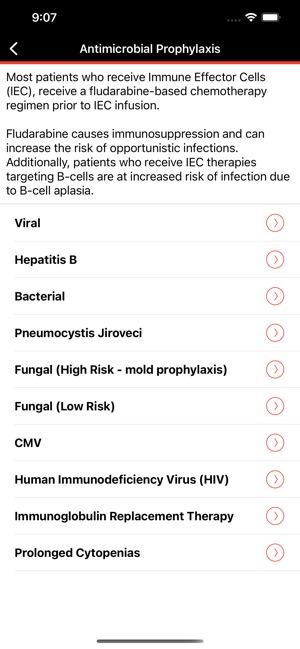
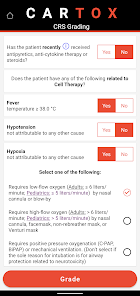
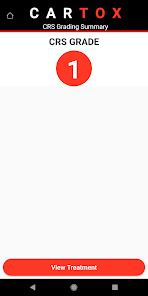
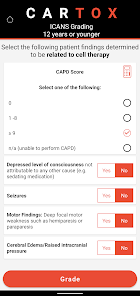
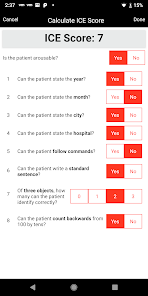
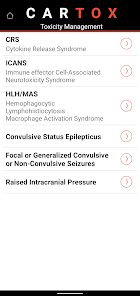
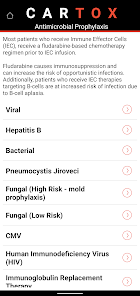
CARTOX is a clinical reference tool that operationalizes consensus-based guidelines for managing cellular therapy toxicities. Developed by the CAR-T-cell-therapy-associated TOXicity (CARTOX) Working Group, the system facilitates the rapid grading of Cytokine Release Syndrome (CRS) and Immune effector Cell-Associated Neurotoxicity Syndrome (ICANS). The interface presents structured algorithms for toxicity management and delivers recommendations for appropriate antimicrobial prophylaxis, integrating evidence-based protocols directly into the clinical workflow for patients receiving immune effector cell therapy.
Evidence & Research Context
- The app's grading and management algorithms are derived from the recommendations of the multi-institutional, multi-disciplinary CARTOX Working Group, established to address the lack of standardized protocols.
- Associated research highlights that significant immune-mediated toxicities, such as CRS and neurotoxicity, occur in up to one-third of patients undergoing cellular immunotherapy.
- The system is designed to address the clinical need for standardized assessment protocols for CRS and Immune effector Cell-Associated Neurotoxicity Syndrome (ICANS).
Intended Use & Scope
This tool is designed for clinicians (physicians, advanced practice providers, pharmacists, nurses) managing patients receiving immune effector cell therapy. Its primary utility is as a point-of-care reference for the rapid grading and management of CRS and ICANS. The app does not replace independent clinical judgment or institutional protocols and is not a substitute for specialist consultation.
Studies & Publications
Peer-reviewed research associated with this app.
Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy
Morris et al. (2021) · Nature Reviews Immunology
Referenced in academic literature; no direct evaluation of the appChimeric antigen receptor T-cell therapy - assessment and management of toxicities
Neelapu et al. (2019) · Nature Reviews Clinical Oncology
Referenced in academic literature; no direct evaluation of the appApp Information
Category
Evidence Profile
Published in academic literature
Platforms
Updated
Sep 2022
© 2025 University of Texas MD Anderson Cancer Center