Contraception
Published in academic literature
App Summary
App Screenshots












Detailed Description
Functionality & Mechanism
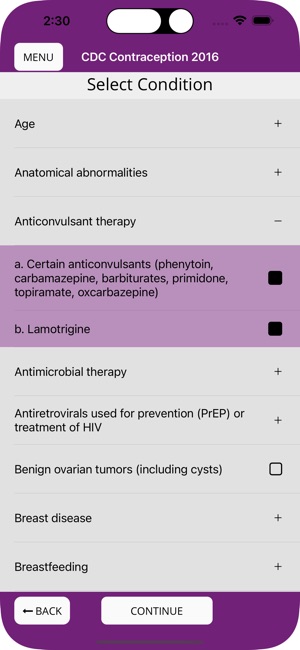
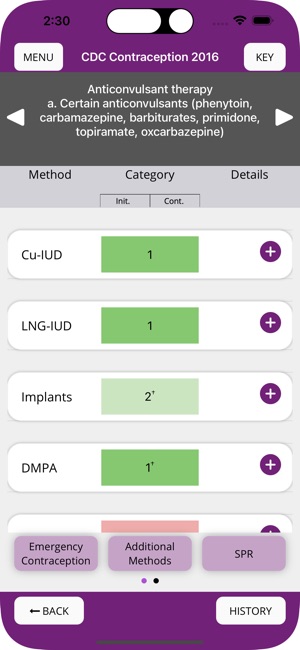
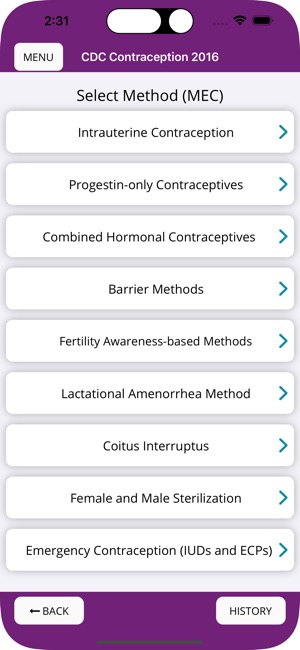
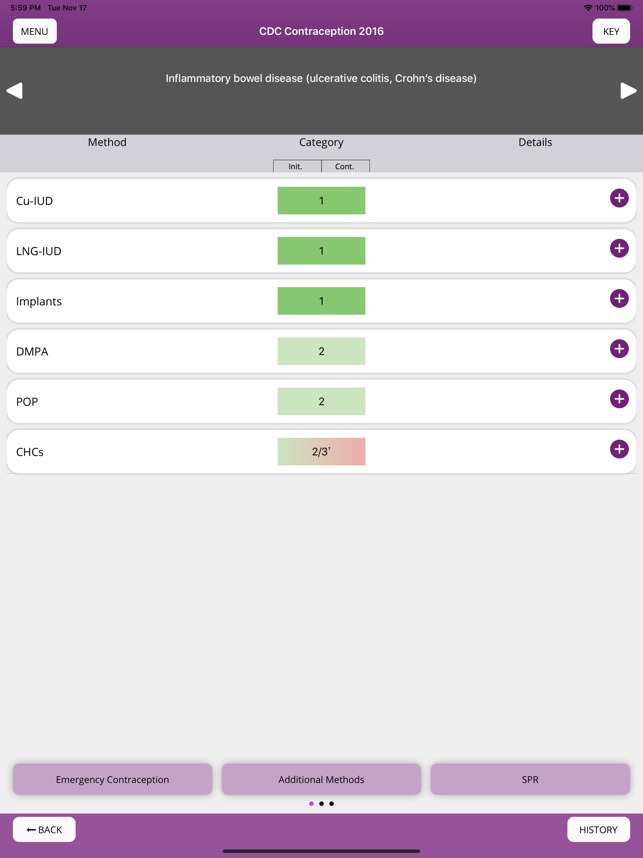
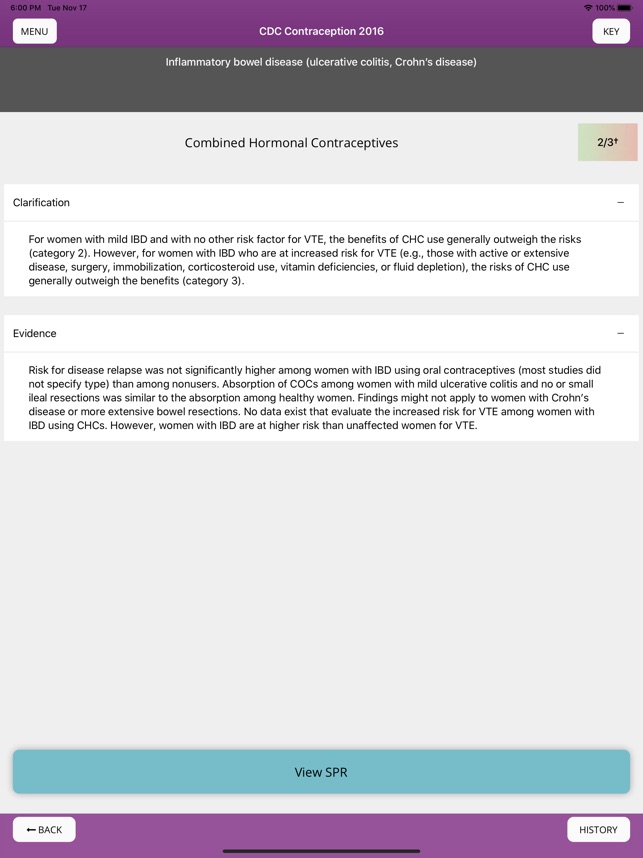
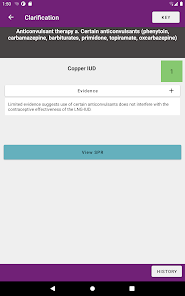
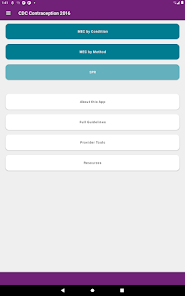
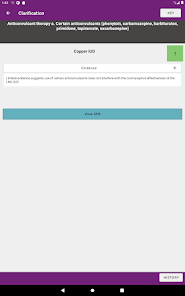
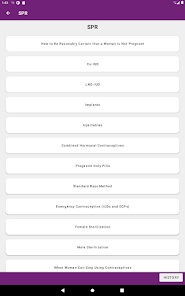
Developed by the Centers for Disease Control and Prevention (CDC), this application translates two key clinical guidelines—the U.S. Medical Eligibility Criteria (MEC) and Selected Practice Recommendations (SPR)—into an accessible reference tool. The interface allows providers to select from over 60 patient characteristics or medical conditions, or to query specific clinical practice situations. The system then presents evidence-based recommendations for contraceptive method selection and management, facilitating rapid access to guidance during clinical encounters.
Evidence & Research Context
- The app's recommendations are directly derived from the CDC's U.S. Medical Eligibility Criteria for Contraceptive Use, which is based on systematic evidence reviews and expert consultation.
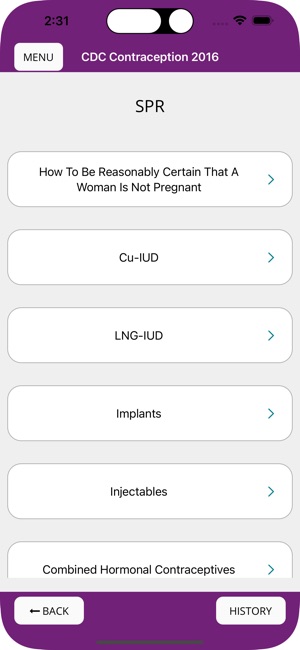
- It also incorporates the CDC's U.S. Selected Practice Recommendations, which address common and complex clinical questions regarding contraceptive initiation and use.
- Associated research details updated eligibility guidance for conditions including chronic kidney disease and systemic lupus erythematosus, and for new contraceptive formulations.
- The source guidelines also provide new recommendations for clinical scenarios such as managing bleeding irregularities with implants and the self-administration of injectable contraception.
Intended Use & Scope
This tool is intended for healthcare providers as a clinical reference for contraceptive counseling and management. Its primary utility is to provide rapid access to evidence-based guidance during patient encounters. The application is not a substitute for individualized clinical judgment and does not provide direct medical advice for patients, who should always consult their provider for care.
Studies & Publications
Peer-reviewed research associated with this app.
U.S. Medical Eligibility Criteria for Contraceptive Use, 2024
Nguyen et al. (2024) · MMWR Recommendations and Reports
Referenced in academic literature; no direct evaluation of the appU.S. Selected Practice Recommendations for Contraceptive Use, 2024
Curtis et al. (2024) · MMWR Recommendations and Reports
Referenced in academic literature; no direct evaluation of the appIn the Media
Contraception App
The CDC developed the Contraception app to help health care providers counsel patients about contraceptive method choice and use, based on CDC Contraceptive Guidance for Health Care Providers. The app covers more than 60 characteristics or medical conditions and numerous clinical situations to serve as clinical guidance. The CDC emphasizes that providers should always consider individual clinical circumstances of each person seeking contraception services.
U.S. MEC and U.S. SPR Provider Tools
The CDC launched the Contraception app in 2024 to provide healthcare providers with easy-to-use contraception guidance, incorporating updated recommendations from the 2024 U.S. MEC and U.S. SPR. The app serves as a comprehensive reference tool that includes provider resources for contraceptive timing, missed dose protocols, and management of complications like bleeding irregularities. The application is available for both iOS and Android operating systems.
Addressing maternal health inequities: Insights from CDC's Wanda Barfield
The CDC's division of Reproductive Health developed the Maternal Mortality Review Information Application (MMRIA) to address significant racial disparities in maternal mortality, using data-driven surveillance to help providers understand factors surrounding maternal deaths. Director Wanda Barfield emphasized that "improving maternal care in the United States is a collective effort" and highlighted the Hear Her campaign, which focuses on urgent maternal warning signs with particular attention to Black and Alaskan Native mothers. The initiative addresses the critical need to reduce pregnancy-related mortality rates that are 3-fold higher in Black women and 2-fold higher in Alaskan Native women compared to White counterparts.
Reproductive Health Access Project
The Centers for Disease Control and Prevention released updated U.S. Medical Eligibility Criteria for Contraceptive Use and U.S. Selected Practice Recommendations for Contraceptive Use on August 6, 2024, to reduce unnecessary barriers to contraceptive care. The updates reflect "an important shift in the framing of contraceptive decision-making, calling out the importance of patient autonomy, patient-centeredness, and shared decision-making." Notable changes include adding chronic kidney disease recommendations and reclassifying combined hormonal contraceptives for sickle cell disease from MEC 2 to MEC 4 due to thrombosis risk.
U.S. Selected Practice Recommendations for Contraceptive Use, 2024
The CDC developed updated U.S. Selected Practice Recommendations for Contraceptive Use in 2024 to address complex issues regarding contraceptive method initiation and use, following a review of scientific evidence and expert consultation in Atlanta during January 2023. The recommendations include notable updates for intrauterine device placement medications, bleeding irregularities during implant use, testosterone use and pregnancy risk, and self-administration of injectable contraception. The guidelines aim to "remove unnecessary medical barriers to accessing and using contraception and to support the provision of person-centered contraceptive counseling and services in a noncoercive manner."
U.S. Medical Eligibility Criteria for Contraceptive Use, 2024
The CDC developed updated Medical Eligibility Criteria for Contraceptive Use to remove unnecessary medical barriers to accessing contraception, using evidence-based recommendations reviewed by national experts in Atlanta during January 25-27, 2023. The 2024 guidelines include notable updates such as new recommendations for persons with chronic kidney disease and inclusion of new contraceptive methods including updated doses of oral contraceptives and vaginal pH modulators. These recommendations aim to "support the provision of person-centered contraceptive counseling and services in a noncoercive manner" for healthcare providers.
App Information
Category
Evidence Profile
Published in academic literature
Platforms
Updated
Aug 2024
© 2025 US Centers for Disease Control and Prevention