ehive Studies
Published in academic literature
App Summary
App Screenshots
Detailed Description
Functionality & Mechanism
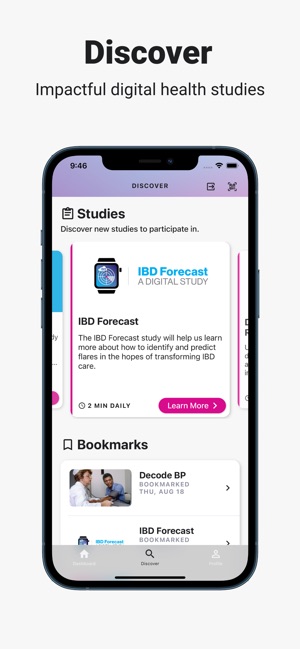
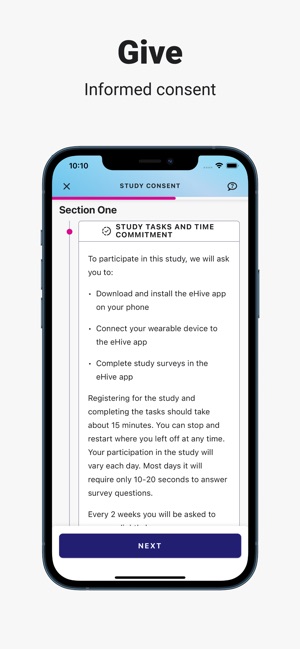
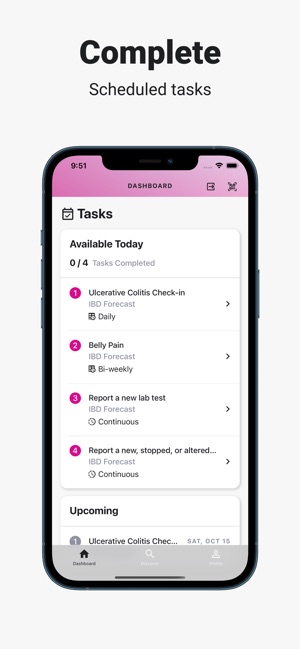
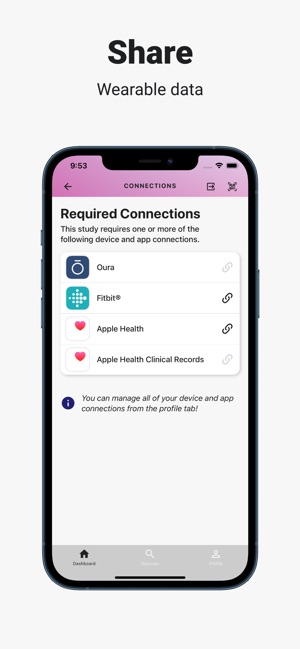
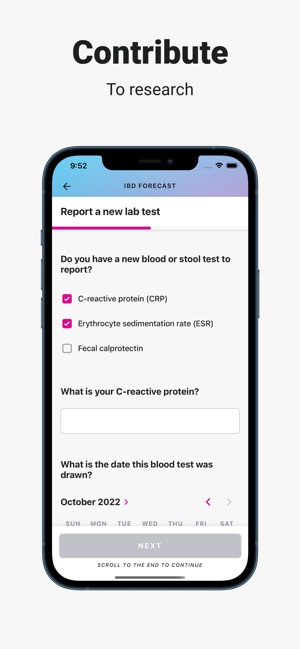
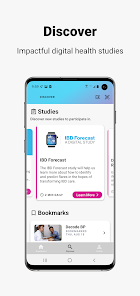
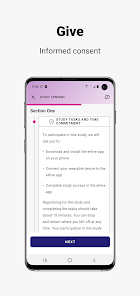
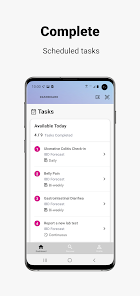
Developed by the Icahn School of Medicine at Mount Sinai, ehive Studies is a centralized digital health platform for longitudinal research. The system facilitates e-consent and subsequent multimodal data collection through a participant-facing mobile interface. Sessions involve study-specific activities such as responding to questionnaires, viewing informational videos, or syncing data from connected wearable devices. The platform leverages automated, light-touch engagement methods to promote high degrees of participant retention for long-term data acquisition.
Evidence & Research Context
- The platform's technical development, e-consent architecture, and data infrastructure are detailed in a published study protocol.
- Since its 2020 launch, the platform has been utilized to host multiple studies, including observational cohorts and clinical trials, enrolling approximately 1,484 participants across 36 U.S. states.
- The system integrates with electronic health records for large-scale recruitment and uses validated methods to sustain participant engagement over time.
Intended Use & Scope
ehive Studies is designed for clinicians and researchers to conduct remote digital health studies and collect longitudinal, real-world data. The platform's primary utility is as a research tool for data acquisition. It does not provide clinical diagnoses, medical advice, or therapeutic interventions. All participation is governed by study-specific protocols approved by an Institutional Review Board.
Studies & Publications
Peer-reviewed research associated with this app.
Development of the ehive Digital Health App: Protocol for a Centralized Research Platform
Hirten et al. (2023) · JMIR Research Protocols
Research study planned or in progress; results not yet availableApp Information
Developer
Mount Sinai Health SystemCategory
Evidence Profile
Published in academic literature
Platforms
Updated
Mar 2026
© 2025 Mount Sinai Health System