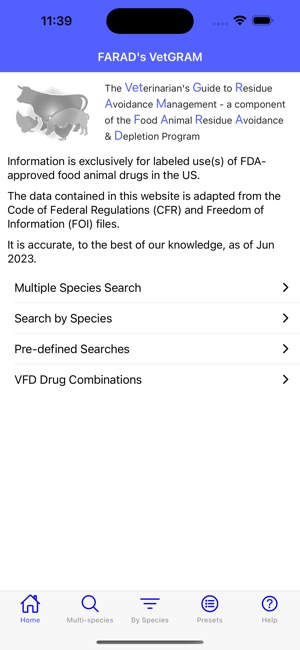
FARAD's VetGRAM
Published in academic literature
App Summary
App Screenshots



Detailed Description
Functionality & Mechanism
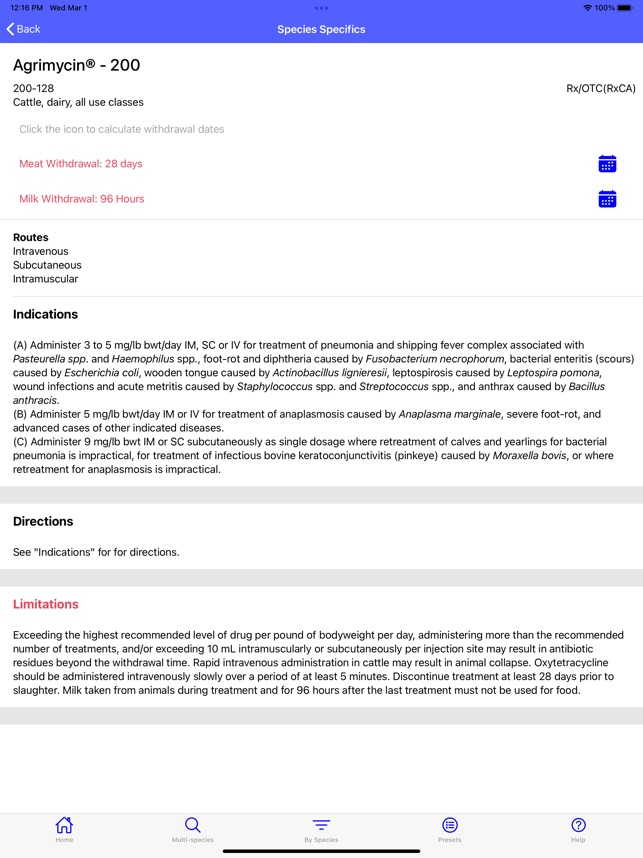
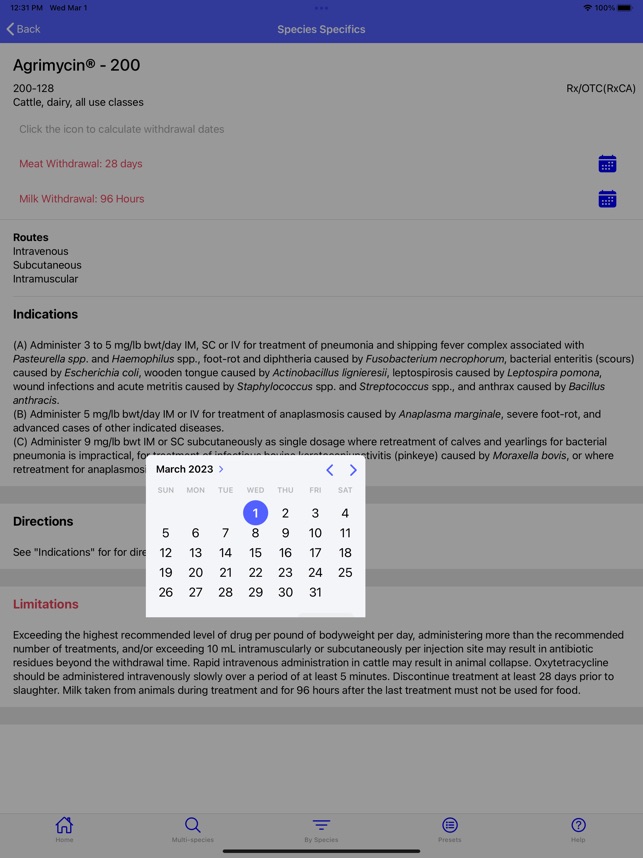
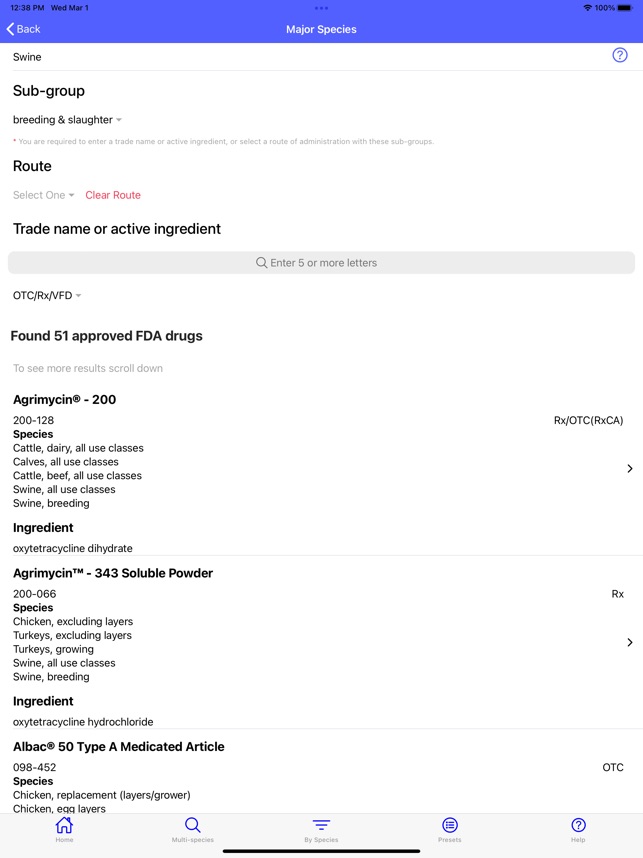
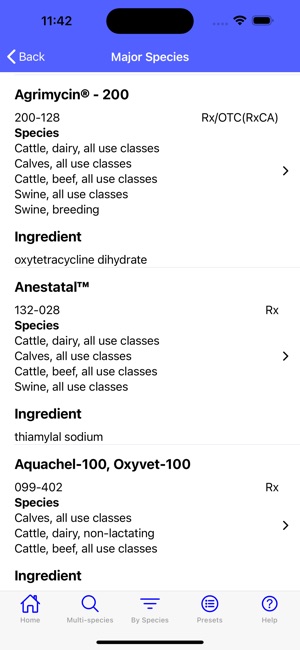
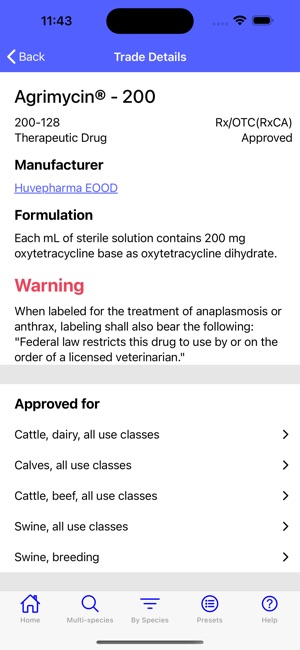
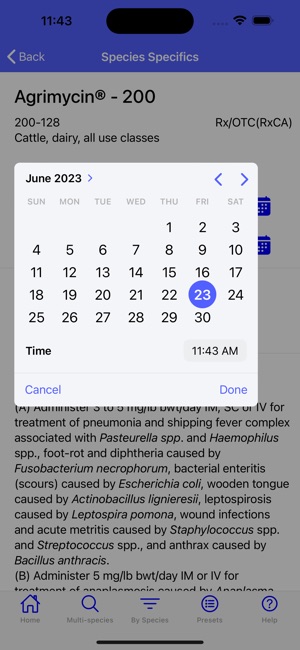
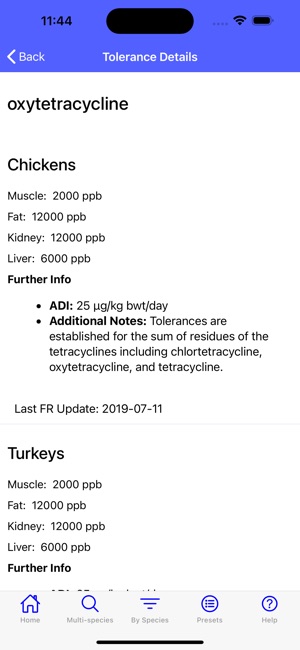
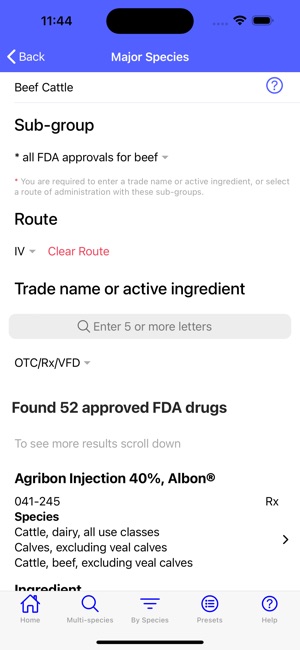
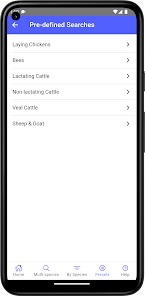
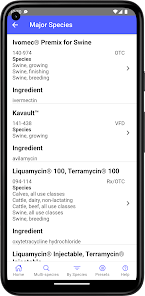
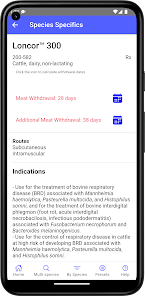
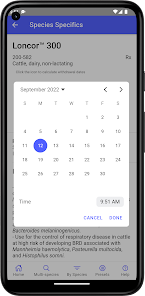
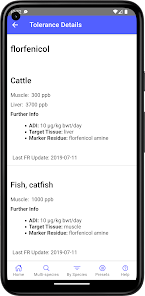
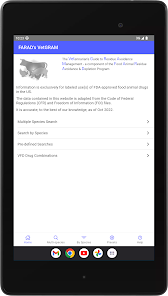
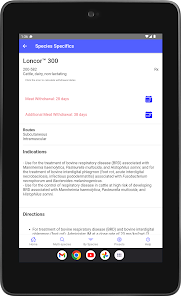
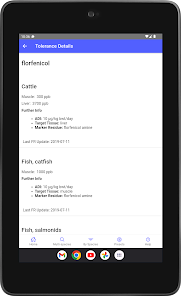
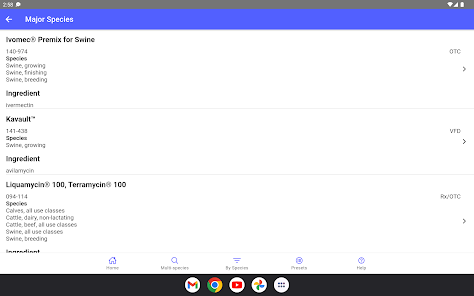
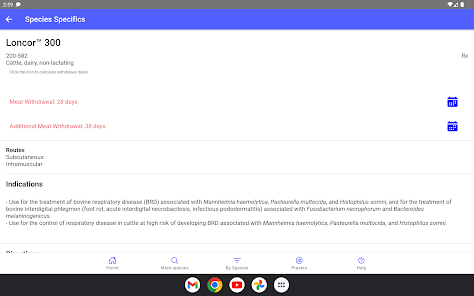
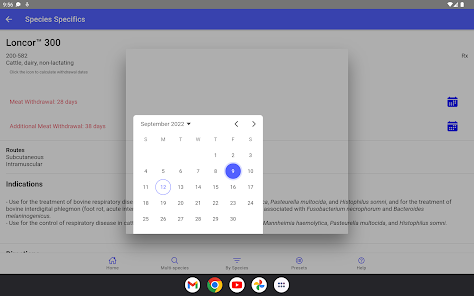
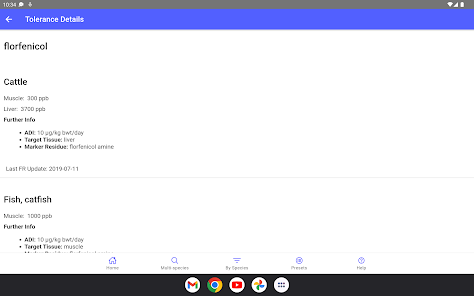
Developed by the University of Florida for the Food Animal Residue Avoidance Databank (FARAD) program, VetGRAM is a comprehensive, offline-accessible clinical reference tool. The interface facilitates customized searches for drugs approved in food-producing species, retrieving information on approved uses, government restrictions, and tolerances. A core module calculates the precise date that animals or their products can enter the food supply, based on FDA-mandated withdrawal intervals following administration at the approved label dose.
Evidence & Research Context
- The underlying Food Animal Residue Avoidance Databank (FARAD) integrates regulatory and pharmacologic information to mitigate drug and chemical residue risks in food animals.
- The databank's scientific methodology leverages statistical analysis of published pharmacokinetic data to produce novel estimates for drug depletion rates in target species.
- Associated research details the program's development of mechanistic, physiologically based pharmacokinetic (PBPK) modeling to refine withdrawal interval predictions, particularly for extralabel drug use.
- The FARAD program, which supports the app's data, is funded by the United States Department of Agriculture (USDA) National Institute of Food and Agriculture (NIFA).
Intended Use & Scope
VetGRAM is designed for licensed veterinarians and veterinary professionals managing pharmacotherapy in food-producing animals. Its primary utility is as a point-of-care reference to verify FDA-approved drug uses and ensure compliance with mandatory withdrawal intervals. The app does not provide recommendations for extralabel drug use; such cases necessitate direct consultation with FARAD.
Studies & Publications
Peer-reviewed research associated with this app.
Integration of Food Animal Residue Avoidance Databank (FARAD) empirical methods for drug withdrawal interval determination with a mechanistic population-based interactive physiologically based pharmacokinetic (iPBPK) modeling platform: example for flunixin meglumine administration
Li et al. (2019) · Archives of Toxicology
Describes the research-driven development of this appFood Animal Residue Avoidance Databank (FARAD): An Automated Pharmacologic Databank for Drug and Chemical Residue Avoidance
Riviere et al. (1986) · Journal of Food Protection
Referenced in academic literature; no direct evaluation of the appApp Information
Developer
University of FloridaCategory
Evidence Profile
Published in academic literature
Platforms
Updated
Apr 2024
© 2025 University of Florida