iChoose Kidney - Educational
Studied in clinical trials · Mixed evidence
App Summary
App Screenshots





Detailed Description
Functionality & Mechanism
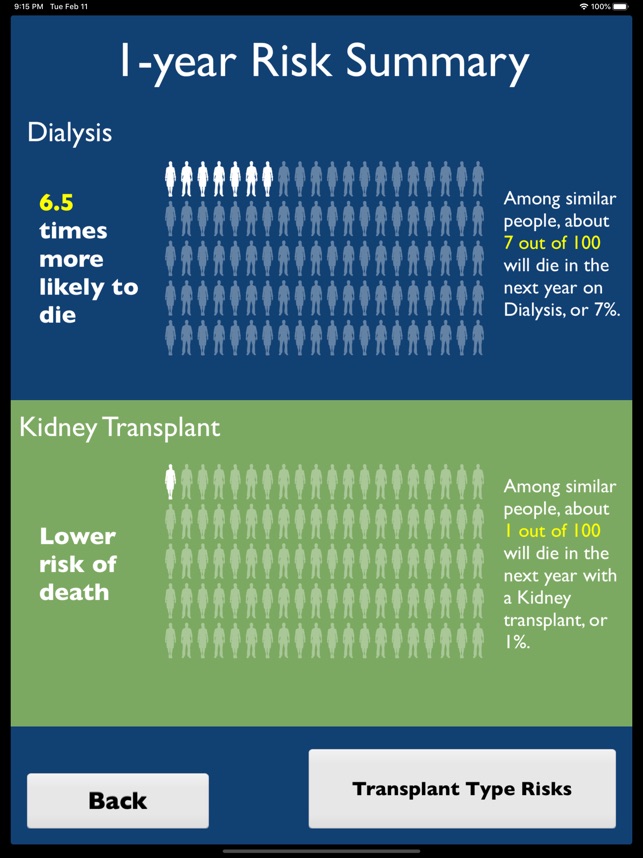
The iChoose Kidney decision aid is a clinical support tool for mobile and web platforms, designed to be used during patient encounters. The system leverages validated predictive models to deliver individualized 1- and 3-year mortality risk estimates for patients with end-stage renal disease (ESRD). The interface captures key patient characteristics—including demographics, comorbidities, and dialysis modality—to generate a comparative visualization of survival outcomes for dialysis versus kidney transplantation, thereby facilitating a shared patient-provider discussion on treatment options.
Evidence & Research Context
- A randomized controlled trial (N=443) demonstrated that the decision aid significantly improved patient knowledge of transplant options compared to standard education alone (mean knowledge score change: 1.1 vs. 0.4; P < .0001).
- The same trial determined that use of the tool did not independently increase patient access to transplantation (defined as waitlisting, living donor inquiries, or transplantation).
- The tool's predictive algorithms were developed and validated using a national cohort of over 1.3 million patients from the U.S. Renal Data System (USRDS).
- Validation of the 3-year mortality models demonstrated moderate discriminatory ability (c-statistic ≈ 0.70), and the models have been systematically updated to incorporate more recent data and additional predictors.
Intended Use & Scope
This system is intended for use by clinicians—including nephrologists, primary care physicians, nurses, and social workers—as an adjunct tool during patient consultations. Its primary scope is to facilitate shared decision-making by visualizing comparative mortality risks. The tool provides population-based risk estimates and does not replace individualized clinical assessment, prognostication, or comprehensive patient counseling.
Studies & Publications
Peer-reviewed research associated with this app.
iChoose Kidney for Treatment Options: Updated Models for Shared Decision Aid
Gander et al. (2018) · Transplantation
Describes the research-driven development of this appEffect of the iChoose Kidney decision aid in improving knowledge about treatment options among transplant candidates: A randomized controlled trial
Patzer et al. (2018) · American Journal of Transplantation
Improved patient knowledge about transplant options but did not increase transplantation rates.
App Information
Developer
Emory UniversityCategory
Evidence Profile
Studied in clinical trials · Mixed evidence
Platforms
Updated
Feb 2020
© 2025 Emory University
Tags
Developer Links
Privacy PolicyiChoose Kidney - Educational
Free