MAQI2 Anticoagulation Toolkit
Published in academic literature
App Summary
App Screenshots
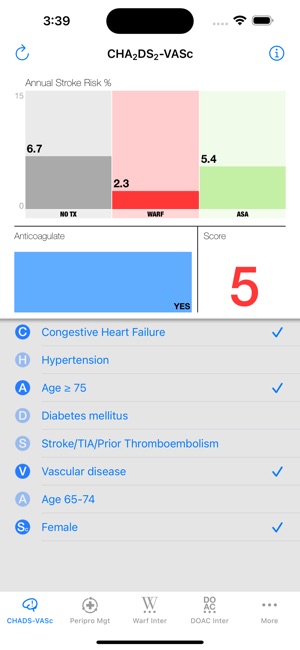

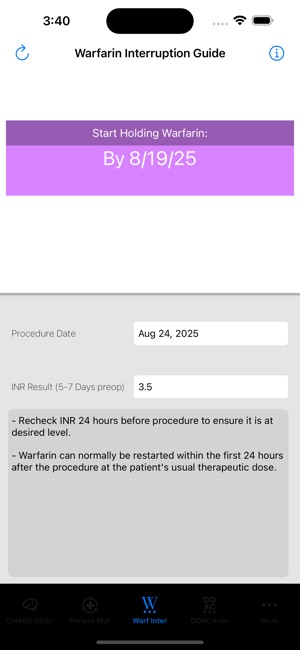
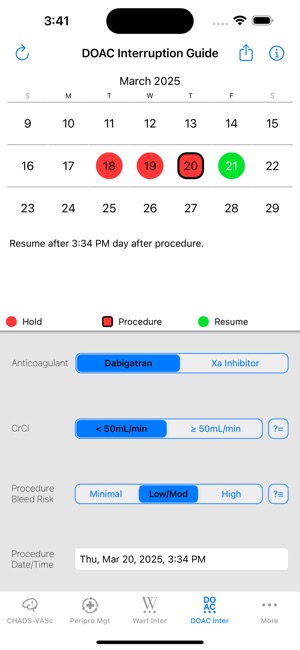
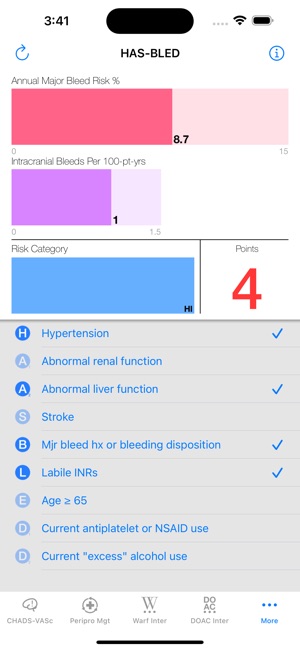
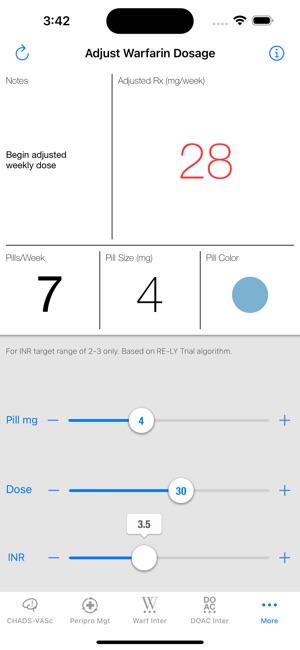
Detailed Description
Functionality & Mechanism
Developed by the Michigan Anticoagulation Quality Improvement Initiative (MAQI2), this clinical toolkit serves as a point-of-care reference for managing patients on anticoagulation therapy. The interface delivers content derived from established clinical guidelines and consortium-led research. Clinicians can access synthesized recommendations and protocols to support decision-making for complex patient scenarios, including dosing adjustments, risk factor management, and therapy transitions. The system is designed to facilitate safe and effective anticoagulation management in various clinical practice settings.
Evidence & Research Context
- The app's development was informed by survey research (N=136) indicating that patients with lower time in therapeutic range (TTR) were receptive to using a mobile support tool for their warfarin therapy.
- Its content is grounded in extensive research from the MAQI2 consortium, which has published observational studies identifying predictors of warfarin discontinuation (N=734) and modifiable risk factors for upper GI hemorrhage (N=6,907).
- As a product of a statewide quality improvement collaborative, the toolkit is designed to translate evidence-based findings into clinical practice to enhance patient safety and standardize care protocols.
Intended Use & Scope
This toolkit is designed for healthcare practitioners, including physicians, pharmacists, and nurses, who manage patients on anticoagulation therapies. Its primary utility is as a clinical reference and decision-support resource. The tool is an adjunct to, not a substitute for, comprehensive clinical judgment, direct patient evaluation, and adherence to institutional protocols. It is not intended for direct use by patients.
Studies & Publications
Peer-reviewed research associated with this app.
Missed opportunities to prevent upper GI hemorrhage: The experience of the Michigan Anticoagulation Quality Improvement Initiative
Kurlander et al. (2019) · Vascular Medicine
Anticoagulation clinics identified opportunities to reduce bleeding risk by deprescribing unnecessary antiplatelet drugs and prescribing protective medications.Discontinuation of Warfarin Therapy for Patients With Atrial Fibrillation: The Michigan Anticoagulation Quality Improvement Initiative Experience
Barnes et al. (2017) · JAMA Cardiology
Tracked warfarin discontinuation rates among atrial fibrillation patients.In the Media
MAQI2 Anticoagulation Toolkit App, essential for treating patients requiring anticoagulants
The University of Michigan developed MAQI2 Anticoagulation Toolkit to help clinicians answer difficult questions and make decisions regarding anticoagulation therapy, addressing the complex treatment needs of patients with conditions like atrial fibrillation. The app provides essential guidance for treating patients requiring anticoagulants, particularly given that atrial fibrillation affects an estimated 2.2 million people in the U.S. and is associated with a 5-fold increased risk for stroke.
Michigan Anticoagulation Quality Improvement Initiative Offers Provider & Patient Anticoagulation Toolkits
The MAQI2 collaborative developed the MAQI2 Anticoagulation Toolkit to help providers manage anticoagulation patients more safely and effectively, using a downloadable collection of guidelines, protocols, and a mobile app. The toolkit includes patient education resources in multiple languages and is available at www.anticoagulationtoolkit.org. Thousands of providers and patients across the country are currently using the toolkit.
App Information
Developer
University of MichiganCategory
Evidence Profile
Published in academic literature
Platforms
Updated
Dec 2025
© 2025 University of Michigan
Tags
Developer Links
Privacy PolicyMAQI2 Anticoagulation Toolkit
Free