PainTrek
Validated in clinical trials · Supported by multiple studies
App Summary
App Screenshots




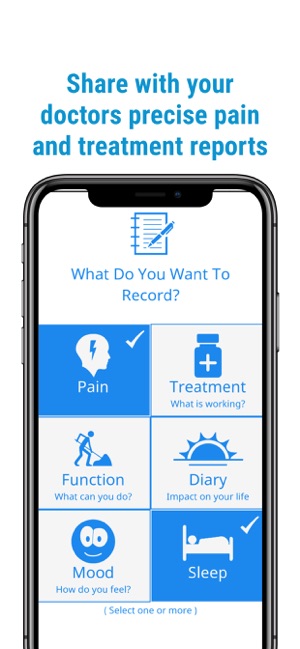

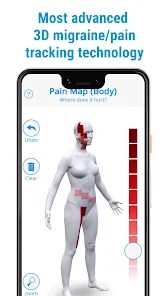


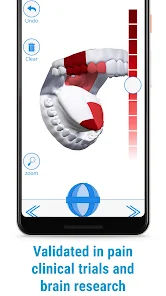
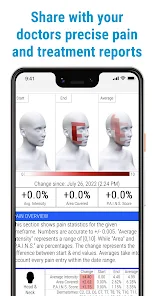
Detailed Description
Functionality & Mechanism
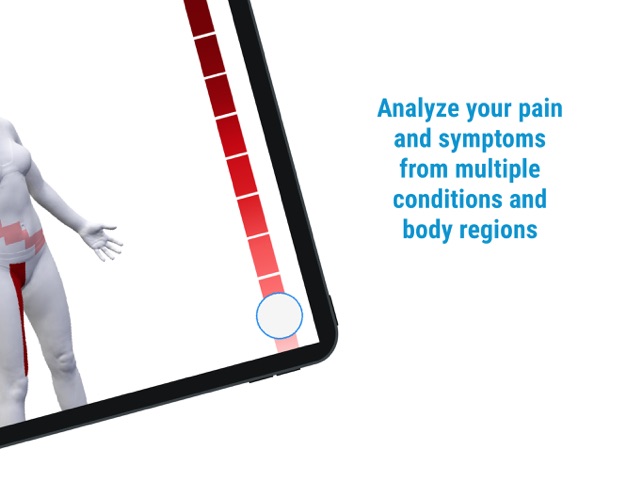
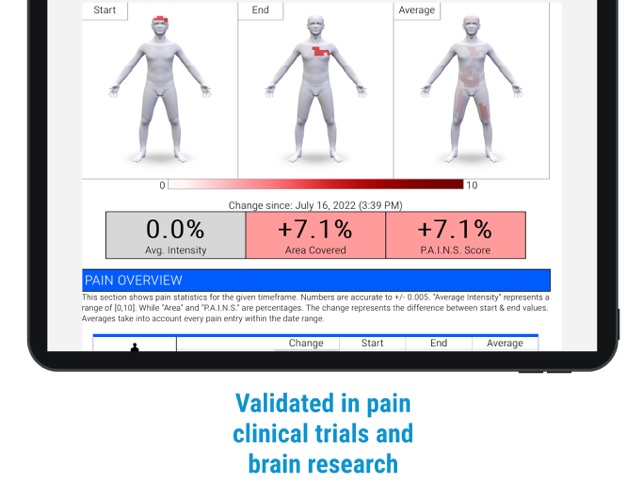
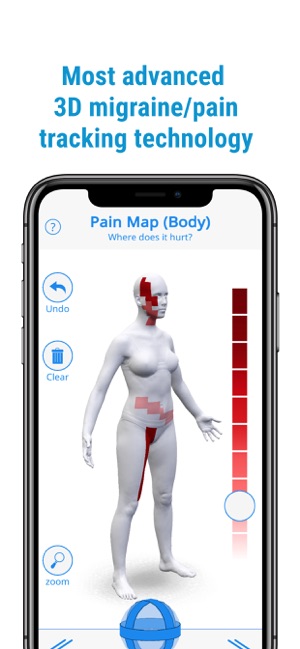
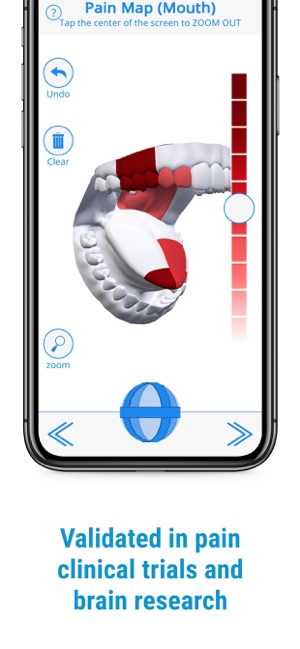
Developed by the H.O.P.E. laboratory at the University of Michigan, PainTrek facilitates high-resolution, longitudinal pain assessment. The system's core interface is a multi-vector, rotational 3D human body model, enabling precise geospatial mapping of pain location, area, and intensity. Sessions involve selecting body regions, assigning validated pain scores, and documenting associated symptoms and triggers. The platform aggregates this data over time, generating shareable, multi-format reports for clinical review or research analysis.
Evidence & Research Context
- A validation study (N=24) of the app's measurement system in patients with temporomandibular disorder (TMD) demonstrated robust long-term reliability (autocorrelations 0.7-0.8) compared to the traditional visual analog scale (VAS) (0.3-0.6).
- The system's composite pain score (PAINS) showed a larger effect size (0.51-0.60 SD) than VAS (0.35 SD), indicating greater sensitivity for detecting treatment effects in clinical trials.
- The study found the app's sensory-discriminative metrics were not significantly influenced by patient mood, in contrast to VAS, which demonstrated a strong correlation with affect scores.
- The app has been successfully utilized as a primary data collection tool in a neuromodulation trial for chronic TMD, capturing significant changes in sectional pain metrics post-intervention.
Intended Use & Scope
Designed for researchers, clinicians, and patients, this tool serves as a high-resolution instrument for longitudinal pain monitoring and data collection in clinical or research settings. Its primary utility is for detailed tracking of complex pain conditions. The system is an assessment aid and does not provide diagnostic outputs or treatment recommendations; professional clinical consultation is required.
Studies & Publications
Peer-reviewed research associated with this app.
Effect of High-Definition Transcranial Direct Current Stimulation on Headache Severity and Central µ-Opioid Receptor Availability in Episodic Migraine
Silva et al. (2023) · Journal of Pain Research
Showed no overall benefit but improved headache outcomes in higher-frequency migraine patients.
Sensory-Discriminative Three-Dimensional Body Pain Mobile App Measures Versus Traditional Pain Measurement With a Visual Analog Scale: Validation Study
Kaciroti et al. (2020) · JMIR mHealth and uHealth
App measurements reliably captured pain patterns and showed stronger performance than traditional pain scales.
In the Media
PainTrek: A Bold Approach to Chronic Pain Treatment
Dr. Alexandre DaSilva and his colleagues at the University of Michigan developed PainTrek to better track, communicate, and understand chronic pain, integrating neuroimaging and brain stimulation research. Developed with support from U-M's Bold Challenges Initiative and Innovation Partnerships, the app offers patients a nuanced way to report pain and enables physicians to track treatment efficacy. PainTrek addresses the needs of millions of Americans who suffer from chronic pain that limits their productivity and reduces their quality of life.
JMIR mHealth and uHealth - Sensory-Discriminative Three-Dimensional Body Pain Mobile App Measures Versus Traditional Pain Measurement With a Visual Analog Scale: Validation Study
University of Michigan researchers developed PainTrek (originally called GeoPain) to address inaccurate pain interpretation from traditional visual analog scales, using a neuroscience-based 3D body pain mapping approach. The validation study with temporomandibular disorder patients used principal component analysis and correlation methods to compare the app's measurements against traditional VAS and mood scales. The research was published in JMIR mHealth and uHealth in August 2020 through collaboration with multiple institutions including the University of Michigan's Center for Computational Medicine and Bioinformatics.
Migraine Pain App Wins 1st Place in U-M Tech Challenge
Dr. Alexandre DaSilva from the University of Michigan's School of Dentistry developed PainTrek to help migraine and facial pain sufferers precisely record pain location in real time on mobile devices, collaborating with the University's 3D Lab to win first place in a U-M Mobile Apps Challenge. "For 15 years I have been working to try to better understand the mechanisms in the brain that trigger headaches and facial pain disorders," DaSilva said, explaining how the app applies these mechanisms clinically for real-time monitoring. The free application uses touch screen technology with a three-dimensional head model and geographical coordinates to pinpoint pain location and track symptoms for more effective treatment planning.
App Information
Developer
University of MichiganCategory
Evidence Profile
Validated in clinical trials · Supported by multiple studies
Platforms
Updated
Feb 2023
© 2025 University of Michigan