PneumoRecs VaxAdvisor
Published in academic literature
App Summary
App Screenshots





Detailed Description
Functionality & Mechanism
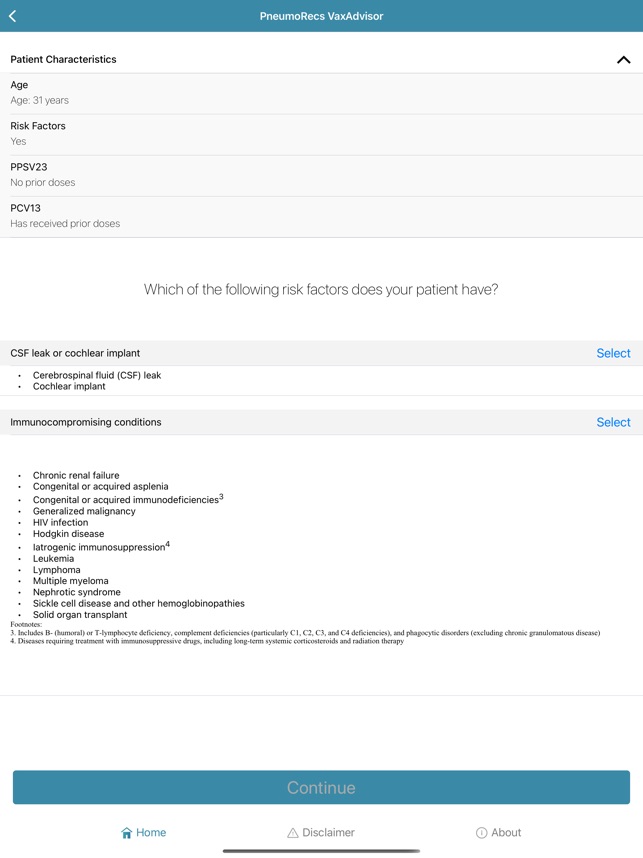
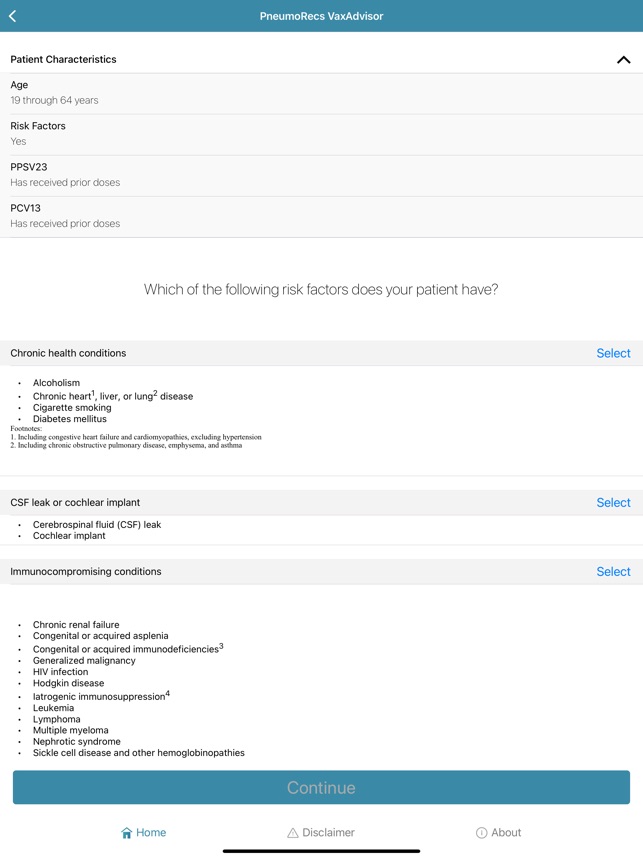
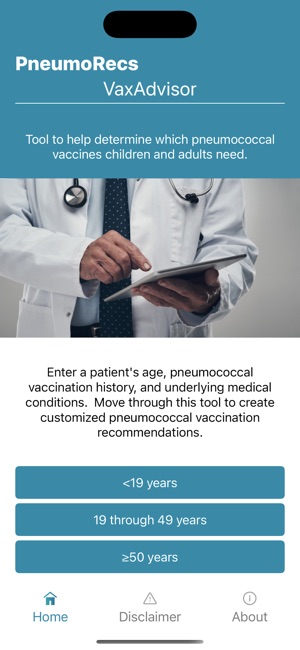
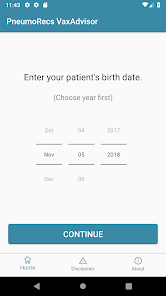
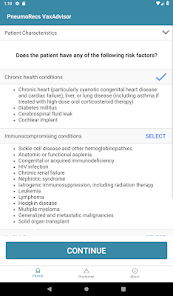
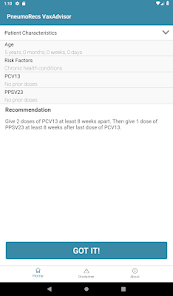
Developed by the Centers for Disease Control and Prevention (CDC) and the Georgia Tech Research Institute, PneumoRecs VaxAdvisor functions as a clinical decision support tool. The system interface prompts for key patient variables including age, pneumococcal vaccination history, and underlying medical conditions. Leveraging this input, the application's algorithm processes the data against established immunization logic. It then generates patient-specific pneumococcal vaccination recommendations, delivering a clear, actionable output for the provider within a single session.
Evidence & Research Context
The tool's recommendation engine is designed to be consistent with U.S. clinical guidance from the Advisory Committee on Immunization Practices (ACIP).
- The associated ACIP guidance recommends a single dose of a pneumococcal conjugate vaccine (PCV) for all vaccine-naïve adults aged 50 years and older.
- The framework accounts for multiple vaccine options, including PCV20 or PCV21 alone, or PCV15 administered in series with PPSV23.
- The system incorporates distinct recommendations for adults aged 19–49 with specific risk conditions for pneumococcal disease.
- It also provides guidance for adults who have previously initiated their pneumococcal vaccination series with PCV13.
Intended Use & Scope
This application is intended for immunization providers and clinicians to facilitate the determination of appropriate pneumococcal vaccination schedules. Its primary utility is as a point-of-care reference tool that translates complex guidelines into specific recommendations. The tool augments clinical judgment but does not replace comprehensive patient assessment or the provider-patient relationship.
Studies & Publications
Peer-reviewed research associated with this app.
Expanded Recommendations for Use of Pneumococcal Conjugate Vaccines Among Adults Aged ≥50 Years: Recommendations of the Advisory Committee on Immunization Practices — United States, 2024
Kobayashi et al. (2025) · MMWR Morbidity and Mortality Weekly Report
Referenced in academic literature; no direct evaluation of the appApp Information
Category
Evidence Profile
Published in academic literature
Platforms
Updated
Aug 2025
© 2025 US Centers for Disease Control and Prevention