SCI Cancer Clinical Trials
Initial evidence from research studies
App Summary
App Screenshots





Detailed Description
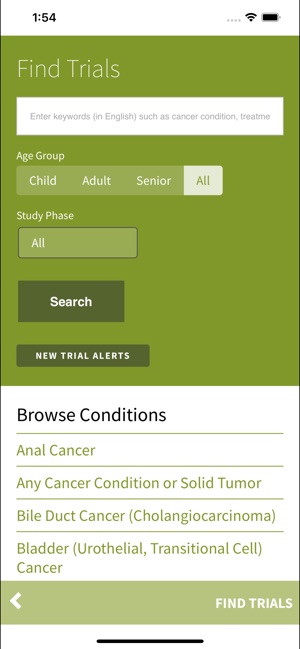
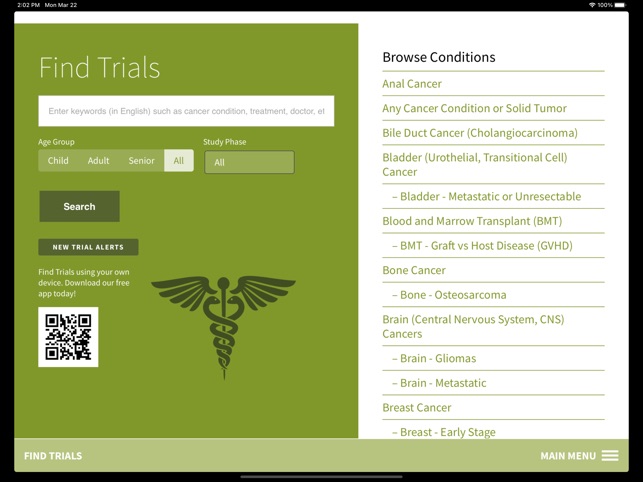
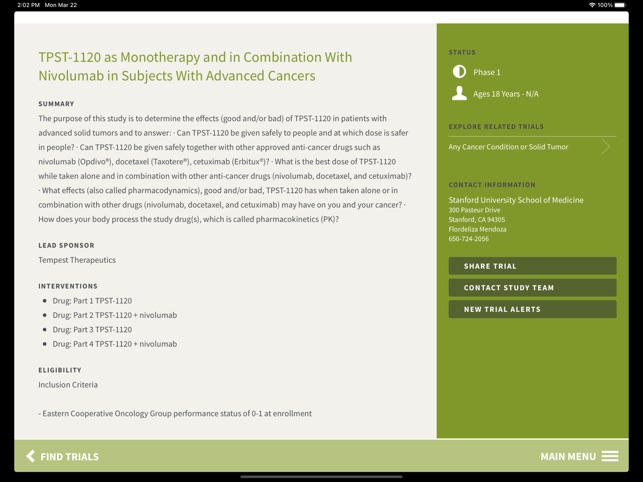
Functionality & Mechanism Developed by the Stanford Cancer Institute, this tool provides a searchable interface for over 250 active cancer clinical trials. The system facilitates trial discovery by cancer condition or keyword search. Core functionality permits users to transmit trial information via email and initiate direct communication with specific trial coordinators or the central Cancer Clinical Trials Office to inquire about participation. The interface is designed for rapid information retrieval and connection to research staff.
Evidence & Research Context
- The app provides public access to trials managed within an institutional framework designed to improve participant screening and enrollment.
- Associated research details the development of a related, EMR-integrated clinical trial matching system for healthcare providers at Stanford.
- Implementation of the provider-facing system demonstrated a significant increase in screening referrals over three years, from 20 to 236 annually.
- The conversion rate for these system-facilitated referrals, defined as subsequent trial enrollment, was documented between 16% and 26%.
Intended Use & Scope This application is intended as a clinical reference tool for patients, caregivers, and clinicians to identify potentially relevant trials at the Stanford Cancer Institute. Its primary utility is for information discovery. The tool does not assess eligibility, provide medical advice, or constitute enrollment. Formal screening must be initiated directly with the trial's research team.
Studies & Publications
Peer-reviewed research associated with this app.
Development and implementation of cancer clinical trial patient screening using an electronic medical record-integrated trial matching system
Bui et al. (2025) · BMJ Health & Care Informatics
Trial screening referrals increased more than tenfold over three years with consistent enrollment conversion.
App Information
Developer
Stanford UniversityCategory
Evidence Profile
Initial evidence from research studies
Platforms
Updated
Apr 2021
© 2025 Stanford University
Tags
Developer Links
Privacy PolicySCI Cancer Clinical Trials
Free