LDL Cholesterol Calculator
Validated in clinical trials · Initial evidence
App Summary
App Screenshots

Detailed Description
Functionality & Mechanism
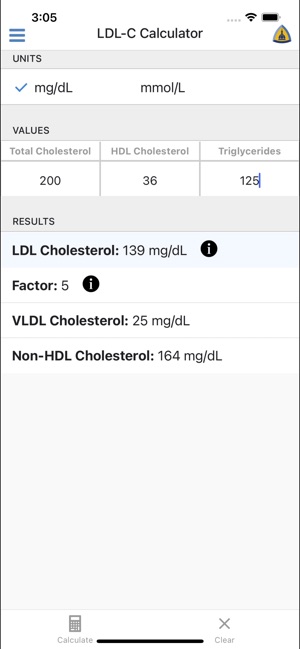
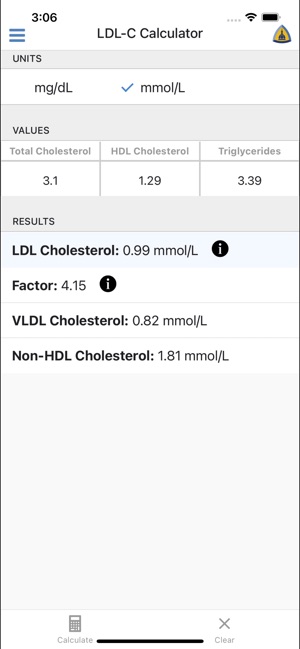
Developed by the Johns Hopkins Ciccarone Center, this tool provides automated calculation of low-density lipoprotein cholesterol (LDL-C). The interface captures three standard inputs: total cholesterol, HDL cholesterol, and triglycerides. It then leverages the Martin-Hopkins method, which applies a personalized factor for the TG:VLDL-C ratio based on the patient's specific lipid values. This dynamic approach contrasts with the fixed-factor calculation of the traditional Friedewald equation, facilitating a more precise and individualized estimation.
Evidence & Research Context
- A large-scale validation study (N=450,303) demonstrated that the Martin-Hopkins method achieved significantly higher concordance (91.7%) with directly measured LDL-C levels compared to the Friedewald equation (85.4%).
- The method substantially improves accuracy for classifying low LDL-C levels (<70 mg/dL), especially in patients with high triglycerides (200-399 mg/dL), where classification accuracy increased from 40.3% to 84.0%.
- Subsequent research has robustly validated the equation in diverse global populations exceeding 5 million individuals, including those with diabetes, kidney disease, and other common comorbidities.
- The calculation method is recommended in clinical practice guidelines from the American Heart Association/American College of Cardiology and other international expert societies.
Intended Use & Scope
This calculator is intended for clinicians and researchers for the accurate estimation of LDL-C. Its primary utility is as a clinical reference tool to guide risk assessment, particularly in patient profiles where traditional formulas are less reliable. The tool's output is for informational purposes and does not replace comprehensive clinical evaluation or the determination of individualized therapeutic plans.
Studies & Publications
Peer-reviewed research associated with this app.
Extensive Evidence Supports the Martin-Hopkins Equation as the LDL-C Calculation of Choice
Grant et al. (2023) · Clinical Chemistry
Referenced in academic literature; no direct evaluation of the appComparison of a novel method vs the Friedewald equation for estimating low-density lipoprotein cholesterol levels from the standard lipid profile
Martin et al. (2013) · JAMA
New estimation method accurately matched directly measured cholesterol values 92% of the time versus 85% for standard equation.
In the Media
Martin/Hopkins Method to Calculate LDL or 'Bad' Cholesterol Outperforms Other Equations, Study Shows
Johns Hopkins researchers developed the Martin/Hopkins method to calculate LDL cholesterol with higher accuracy than existing equations, analyzing data from over 5 million patients. The study found that the Martin/Hopkins algorithm correctly classified 89.6% of patients' LDL cholesterol values, outperforming the previous gold standard Friedewald method which achieved 83.2% accuracy. "The biggest concern is that underestimating LDL cholesterol could lead to withholding treatments that would be beneficial for patients," says lead researcher Dr. Seth Martin.
As Medical Apps Multiply, Johns Hopkins Takes the Lead on Standards
Johns Hopkins Medicine launched two review boards to establish high standards for accuracy and clinical value in health-related apps, addressing widespread inaccuracies in the largely unregulated mobile health marketplace. Johns Hopkins cardiologist Seth Martin's 2016 study found that more than 75% of people with hypertensive blood pressure received falsely reassuring information from a popular blood pressure app. "Johns Hopkins is a place that deeply respects the importance of science and evidence to guide what we do," Martin says.
App Makes Calculating LDL Cholesterol Easy for Physicians
Johns Hopkins cardiologists Steven Jones and Seth Martin created LDL Cholesterol Calculator to make their complex Martin-Hopkins calculation method accessible for physicians to accurately assess patients' cholesterol levels using lab results. "The beauty is the app is straightforward," says Martin, noting that "in a matter of seconds, you can get an accurate LDL cholesterol result." The app has been downloaded more than 10,000 times and serves as a free alternative for health facilities that haven't adopted the Martin-Hopkins calculation software used by Johns Hopkins Medicine and Quest Diagnostics.
App Information
Developer
Johns Hopkins UniversityCategory
Evidence Profile
Validated in clinical trials · Initial evidence
Platforms
Updated
Jun 2021
© 2025 Johns Hopkins University
Tags
Developer Links
Privacy PolicyLDL Cholesterol Calculator
Free