MeTime Acupressure
Validated in clinical trials · Initial evidence
App Summary
App Screenshots










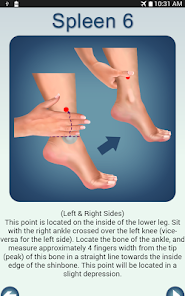
Detailed Description
Functionality & Mechanism
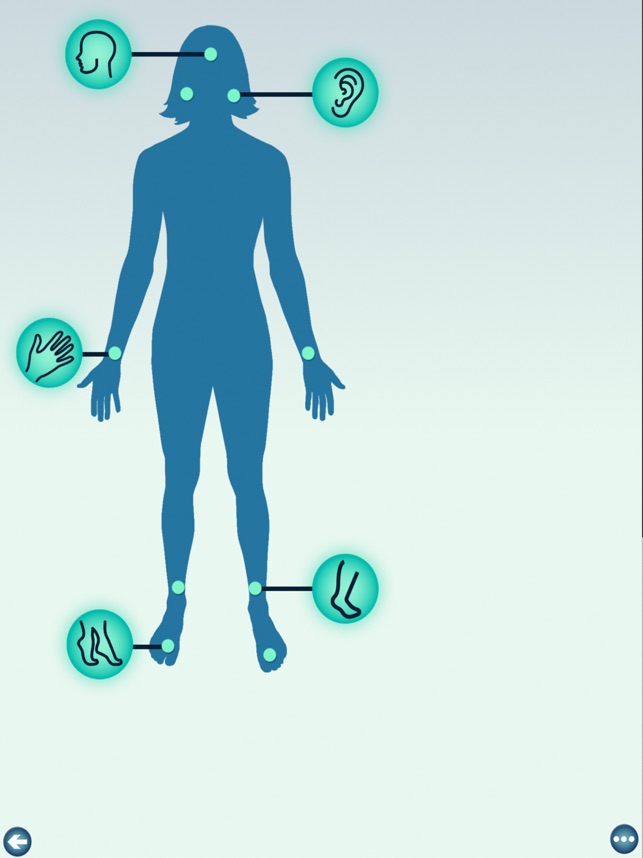
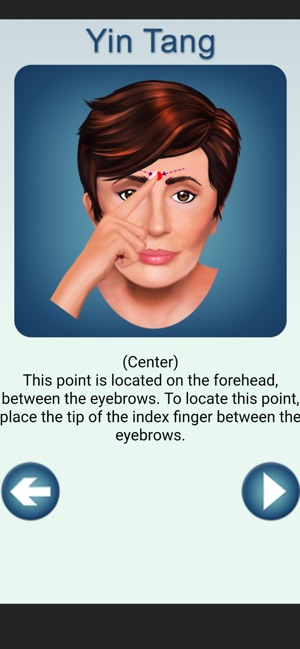
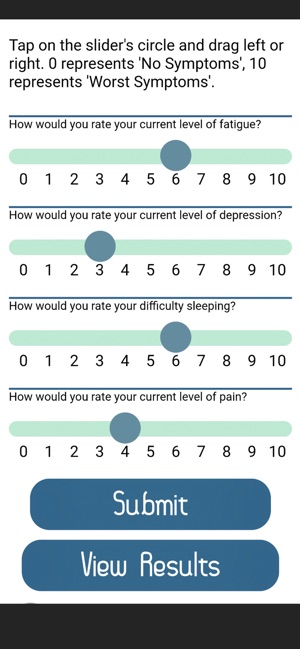
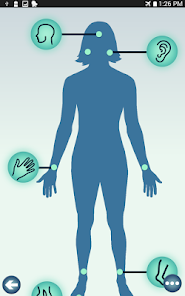
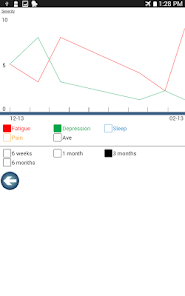
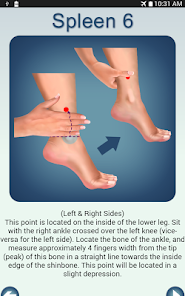
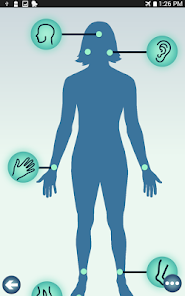
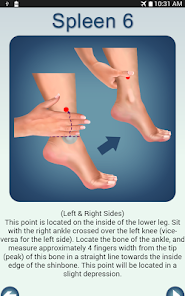
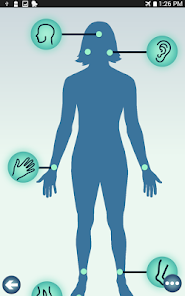
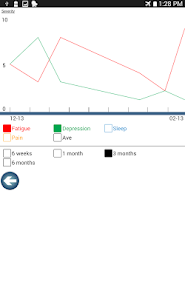
Developed by researchers at the University of Michigan, MeTime Acupressure delivers a pedagogical protocol for self-administered acupressure to manage cancer-related fatigue. The system instructs users on the location and stimulation of specific acupoints. Daily sessions involve applying pressure to 5-7 points for up to three minutes each, consistent with the underlying clinical trial protocol. The interface is structured to guide users through a six-week program designed for sustained symptom management and improved sleep quality.
Evidence & Research Context
- A randomized clinical trial (N=288) of breast cancer survivors with persistent fatigue evaluated the self-acupressure protocol upon which the app is based.
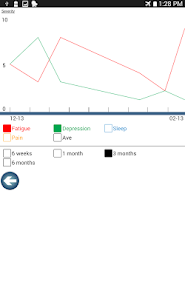
- After six weeks of daily practice, 61-66% of participants in the acupressure arms achieved normal fatigue levels, compared to 31% in the usual care arm.
- The relaxing acupressure protocol also demonstrated significant improvements in sleep quality and overall quality of life compared to usual care.
- The app's methodology is grounded in traditional Chinese medicine principles, with mechanistic studies suggesting acupressure may influence brain connectivity related to sleep and alertness.
Intended Use & Scope
This application is designed for cancer survivors as an adjunct self-management tool for persistent fatigue. Its primary utility is to deliver a structured, evidence-based, non-pharmacologic intervention. The tool does not provide medical advice or replace professional oncological care. Users should consult their healthcare provider to determine if this technique is appropriate for their individual treatment plan.
Studies & Publications
Peer-reviewed research associated with this app.
Investigating Self-Acupressure for Fatigued Ovarian Cancer Survivors
Zick et al. (2019) · Defense Technical Information Center
Referenced in academic literature; no direct evaluation of the appAcupressure for Cancer Related Fatigue
Zick et al. (2018) · The ASCO Post
Referenced in academic literature; no direct evaluation of the appIn the Media
Brain Imaging Study Shows How Acupressure Fights Fatigue in Breast Cancer Survivors
University of Michigan researchers used MRI brain imaging to examine how two types of acupressure affect fatigue in breast cancer survivors, marking the first neuroimaging study of its kind. "What is intriguing about this manuscript is that it shows that the two types of acupressure â stimulating and relaxing â do appear to work via different mechanisms within the brain despite the fact that they both reduced fatigue," says study author Suzanna Zick. The double-blind study examined 19 female breast cancer survivors, focusing on connections between the brainstem and default mode network.
Acupressure Reduces Fatigue in Diagnosed Women
Researchers conducted a randomized clinical trial with 270 breast cancer survivors to test whether self-administered acupressure could reduce persistent cancer-related fatigue, with participants performing daily treatments for 6 weeks. After 6 weeks, 66.2% of women in the relaxing acupressure group and 60.9% in the stimulating acupressure group achieved normal fatigue levels, compared to only 31.3% in the usual care group. The research was published in JAMA Oncology in July 2016.
Acupressure reduced fatigue in breast cancer survivors
University of Michigan researchers developed MeTime Acupressure to help breast cancer survivors reduce persistent fatigue, using traditional Chinese medicine techniques that patients can perform at home. The study published in JAMA Oncology found acupressure reduced fatigue by 27 percent to 34 percent over six weeks, with two-thirds of women achieving normal fatigue levels through relaxing acupressure. "Acupressure is easy to learn and patients can do it themselves," says study author Suzanna Zick, who notes that participants needed only 15 minutes of training to accurately locate pressure points.
App Information
Developer
University of MichiganCategory
Evidence Profile
Validated in clinical trials · Initial evidence
Platforms
Updated
May 2020
© 2025 University of Michigan