UCSF WYZ
Shows promise in preliminary research
App Summary
App Screenshots

Detailed Description
Functionality & Mechanism
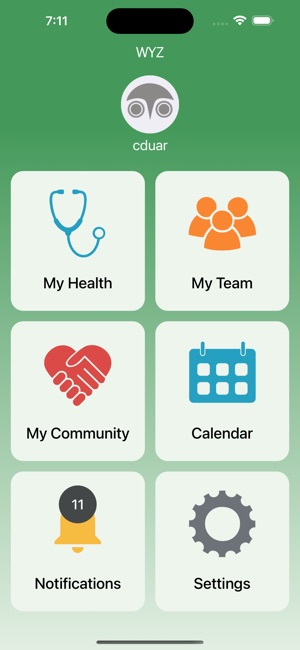
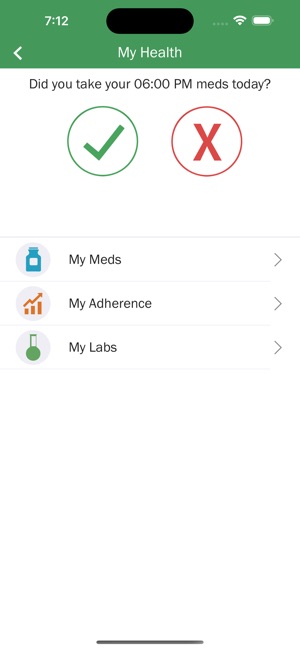
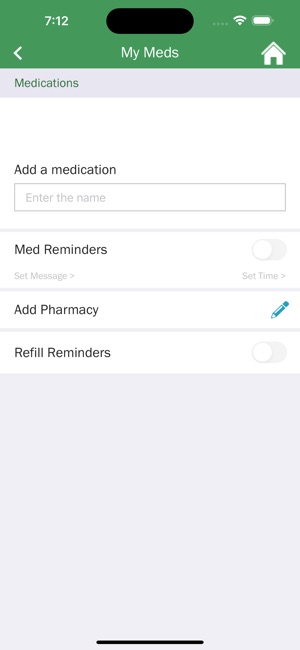
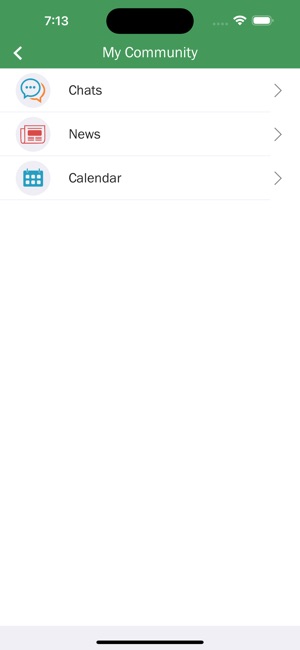
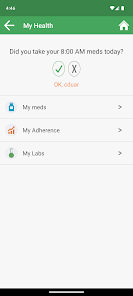
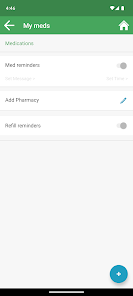
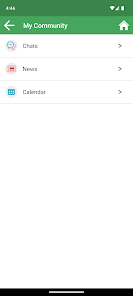
Developed by researchers at the University of California, San Francisco (UCSF), WYZ is a mobile health intervention grounded in the information, motivation, and behavioral skills framework. The system integrates three core modules: 'My Health' facilitates medication and lab result tracking; 'My Team' provides a curated directory of community-based health, housing, and social services; and 'My Community' delivers a secure platform for peer-to-peer communication, medical news, and appointment reminders. The interface was co-designed with a youth advisory board.
Evidence & Research Context
- A preliminary, single-arm pilot study (N=79) evaluated the app's feasibility and acceptability over six months among youth and young adults living with HIV.
- The intervention demonstrated high feasibility, meeting or exceeding predefined benchmarks for recruitment (94%), mean weekly logins (5.3), and weekly peer chat engagement (4.8 posts).
- User-reported acceptability was high, with over 91% of participants favorably rating the app's ease of use, security, and privacy protocols.
- While the study established feasibility, the authors note that further research is required to determine the app's efficacy for improving engagement in care and ART adherence.
Intended Use & Scope
This app is intended for use by youth and young adults living with HIV as an adjunctive tool to support care engagement and self-management. The platform is designed to enhance medication adherence self-efficacy and facilitate access to community resources. It does not provide medical advice and is not a substitute for professional clinical care.
Studies & Publications
Peer-reviewed research associated with this app.
A Mobile Health App (WYZ) for Engagement in Care and Antiretroviral Therapy Adherence Among Youth and Young Adults Living With HIV: Single-Arm Pilot Intervention Study
Saberi et al. (2020) · JMIR Formative Research
Feasibility confirmed — high user satisfaction and engagement among youth and young adults living with HIV.
WYZ: a pilot study protocol for designing and developing a mobile health application for engagement in HIV care and medication adherence in youth and young adults living with HIV
Erguera et al. (2019) · BMJ Open
Research study planned or in progress; results not yet availableApp Information
Category
Evidence Profile
Shows promise in preliminary research
Platforms
Updated
Jan 2026
© 2025 University of California, San Francisco